 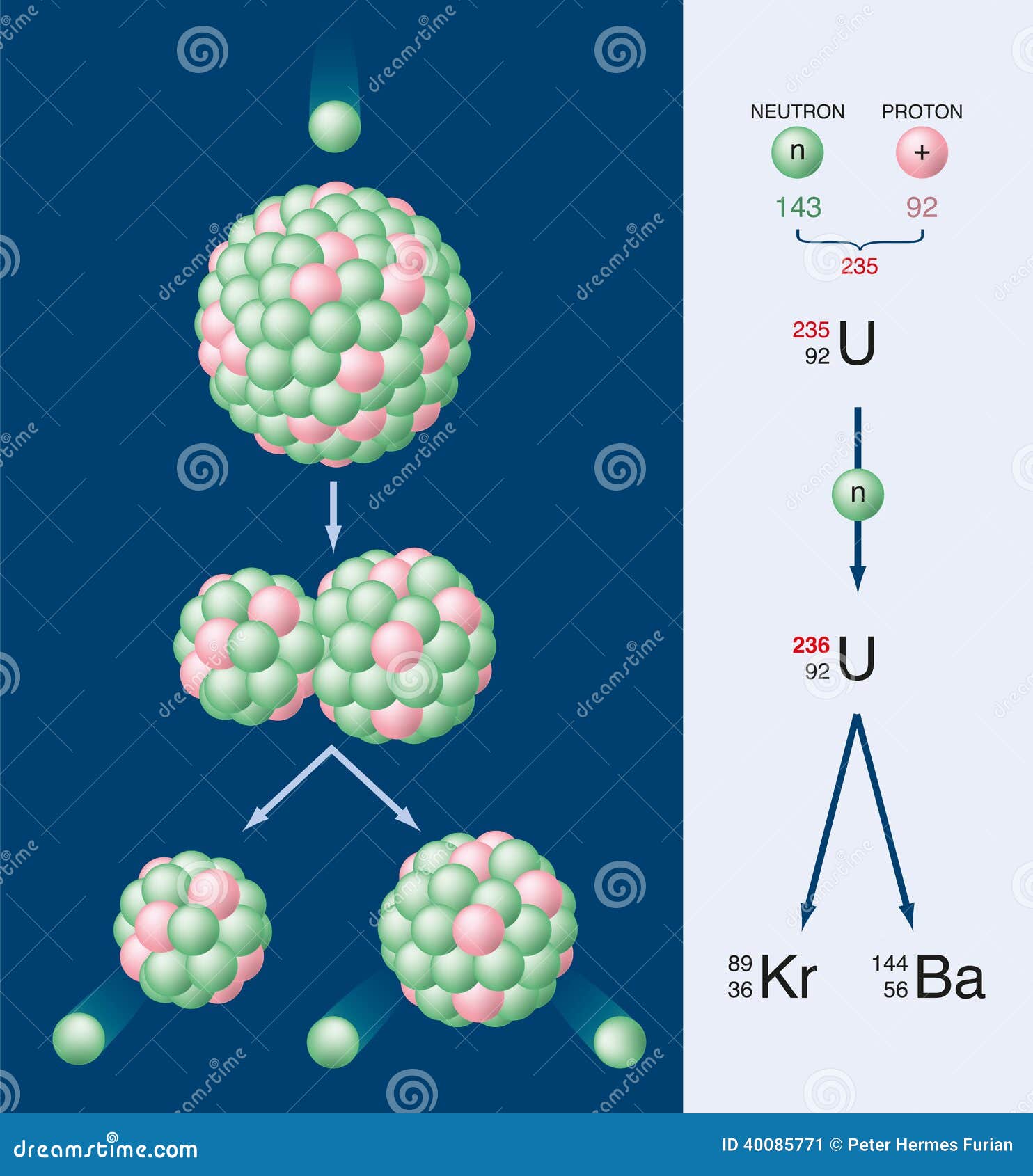
Technetium-99 has a half-life of 211,100 years, but other fission products may have half-lives of about 5 years. Many of the fission products are also radioactive and they decay with a large range of half-lives. The chain reaction in nuclear reactors is controlled to stop it moving too quickly. The chain reaction of splitting a nucleus Even more neutrons are then released, which in turn can split more nuclei. The additional neutrons released may also hit other uranium or plutonium nuclei and cause them to split. In a nuclear reactor, a neutron is absorbed into a nucleus (typically uranium-235). The nuclear symbol for a neutron is \(_\] Chain reaction Nuclear fission is the splitting of a large atomic nucleus into smaller nuclei. Their atoms have relatively large nuclei that are easy to split, especially when hit by neutrons. The original uranium-235 nucleus absorbs a neutron, splits into a krypton-92 nucleus and a barium-141 nucleus, and releases three more neutrons upon splitting. Uranium-238, uranium-235, and thorium-232 have long half-life (238U: 4.5×109 years, 235U: 7.04×108. 1: The nuclear chain reaction is a series of fission processes that sustains itself due to the continuous production of neutrons in each reaction. Uranium recovered from reprocessing of spent nuclear fuel is contaminated with fission products (mainly ruthenium-106 and technetium-99). The rate of decay in spontaneous fission is related to the half-life of the. Properties of the Natural Uranium Isotopes U-234: U-235: U-238: half-life: 244,500 years: 703.8 Uranium or plutonium isotopes are normally used as the fuel in nuclear reactors. Table 1.3) fission produces large nuclear fragments. On average, uranium-235 produces 83 terajoules per kilogram after fission. If neutrons are moving too fast, fission will not occur. Neutrons emitted from such fission can lead to a sustainable chain reaction. This could be taken as a suggestion that geologic storage of radioactive waste is feasible.The absorption of slow neutrons can induce fission in particular nuclei, which are referred to as the fissile nuclei. One of the interesting observations was that the bulk of the fission products seemed to be still in place in their geologic depository after nearly 2 billion years. It is presumed that ground water seeping through the ore served as a natural moderator to slow down the fission neutrons. Nuclear power stations use the fission of uranium-235 to produce thermal. For U-235 (halflife 700 million years) and U-238 (halflife 4.5 billion years), this would give a concentration of about 3% for the U-235 at the time of the reaction. has a half-life of 8 days and, when first injected, its activity is 80 MBq. 235 U is the progenitor of the uranium-235 series, 238 U the progenitor of the uranium-238 series, and 233 U is a member of the neptunium-237 series.Uranium-235 is the most important isotope for nuclear fission. The age estimate from cores in the reactor zones suggest a time frame between 1.7 and 1.9 billion years ago. Uranium plays a major role in the natural radioactive series on the earth. Thus, the nuclear fission observed by Nishina and Kimura is highly symmetric. Nuclear plants provide the world with much of its electricity. An uranium isotope having a half-life of 6.5 d was unknown at that time. This will cause U-235 to become unstable and split up into two light atoms known as fission products. During fission, U-235 atoms absorb loose neutrons. Models of the process suggested sustained fission reactions over a period of about a million years during a time period about two billion years ago. Uranium-235 (U-235) is among the isotopes that fissions easily. Some samples were found with a U-235 concentration as low as 0.29%. Also, fission-produced isotopes of neodynium and samarium were found. the uranium is in large part destroyed in a fraction of a second by fission, and in a nuclear reactor in a few years (by fission and other phenomena), would any of these processes reduce the timing of disappearance of uranium in a, say. In making a bomb or a power plant, then, the first step is to separate the isotopes of uranium from each other. 
This indicated that some of the U-235 had undergone spontaneous nuclear fission at some point in the past. Uranium-235 has a half-life of about 700 million years and a critical mass of about 52. As a chunk of uranium metal contains more uranium-235, it is more likely to undergo nuclear fission. In what is now Gabon in west Africa in 1972, French researchers found a deposit of uranium which had only 0.44% U-235 compared to the normal 0.72%. The uranium-235 first absorbs the neutron to yield uranium-236, and most of these U-236 nuclei split into two fission fragments. Nuclear Fission Concepts The natural fission reactor: Gabon, Africa The most basic fission reaction in nuclear reactors involves the splitting of the nucleus of uranium-235 when it is struck by a neutron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
